Background: Over the past few weeks there has been a shift in the management of critically ill COVID-19 patients. Many seem to have moved away from an intubate early strategy to the use of high flow nasal cannula (HFNC) and noninvasive ventilation (NIV). HFNC and NIV may obviate the need for endotracheal intubation in patients with acute respiratory failure. Mechanical ventilation is not a benign intervention as it has a number of associated complications including ventilator associated pneumonia, excessive sedation, delirium, ICU acquired weakness, as well as ventilator induced lung injury (VILI). NIV can cause lung injury from excessive negative pressure forces. However, mechanical ventilation can cause VILI from excessive positive pressure forces. There is a fine balance of when to use which modality, and when to transition from one modality to another that requires frequent bedside monitoring. NIV has been used successfully for COPD exacerbations and cardiogenic pulmonary edema, but its use in acute hypoxemic respiratory failure is still rather controversial. One of the issues with NIV through a mask device is that higher levels of positive end-expiratory pressure (PEEP), which are often needed in hypoxemic conditions, may lead to mask intolerance and air leaks around the sides of the mask. Helmet NIV may have several advantages over face masks including better tolerability and less air leaks. There is a dearth of evidence comparing these to NIV modalities.
Paper: Patel BK et al. Effect of Noninvasive Ventilation Delivered by Helmet vs Face Mask on the Rate of Endotracheal Intubation in Patients with Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA 2016. PMID: 27179847
Clinical Question: Is noninvasive ventilation (NIV) delivered by helmet better than mask in preventing intubation rate among patients with ARDS?
What They Did:
- Single center randomized clinical trial of 83 consecutive patients with ARDS requiring NIV delivered by face mask for at least 8 hours while in ICU in Chicago
- Planned enrollment of 206 patients
- Trial terminated early due to predefined criteria for efficacy which resulted in 44 patients randomized to the helmet group and 39 to the facemask group
- After 8 hours of facemask NIV, patients randomized to:
- Face Mask NIV (Control)
- Helmet NIV (Intervention)
Outcomes:
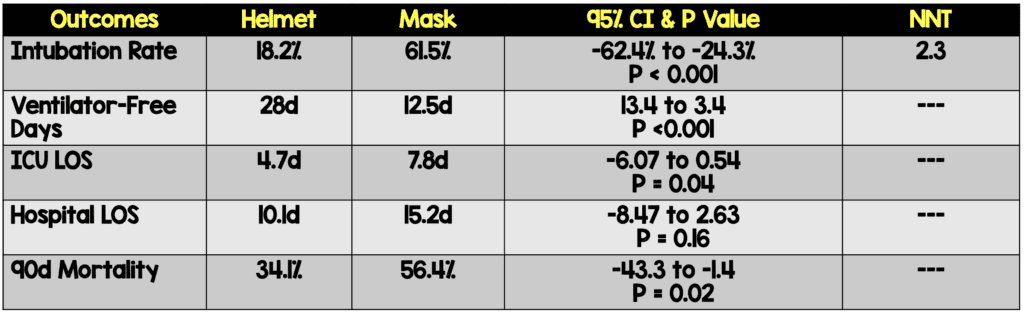
- Primary: Proportion of patient requiring endotracheal intubation
- Secondary:
- 28-day invasive ventilator free days (days alive without mechanical ventilation
- ICU LOS
- Hospital LOS
- Hospital Mortality
- 90d Mortality
- Adverse Events:
- Skin ulceration at seal
- Patient intolerance
- Device complications (helmet deflation)
Inclusion:
- ≥18 years of age
- Required face mask NIV for at least 8 hours for management of ARDS (ARDS defined by Berlin Criteria)
Exclusion:
- Impending cardiopulmonary arrest
- GCS < 8
- Absence of airway protective gag reflex
- Elevated intracranial pressure
- Tracheostomy
- Upper airway obstruction
- Pregnancy
- Refused endotracheal intubation
Results:
- 83 patients enrolled and included in the analysis
- 72% of patients had a PaO2/FiO2 ratio <200
- ≈50% of patients were immunocompromised by virtue of cancer or transplant
- Most Common Reason for Intubation in Face Mask Group = Respiratory Failure (i.e. Tachypnea and hypoxemia)
- Face Mask: 83.3%
- Helmet: 37.5%
- Absolute Difference -45.3; 95% CI -82.5 to -9.1; P = 0.01
- Most Common Reason for Intubation in Helmet Group = Neurologic Failure (i.e. AMS and loss of airway protective reflex)
- Face Mask: 4.2%
- Helmet: 62.5%
- Absolute Difference 58.3; 95% CI 24.8 to 92.8; P = 0.001
- Adverse events included 3 interface-related skin ulcers for each group
- Face Mask: 7.6% (Nose ulcers)
- Helmet: 6.8% (Neck ulcers)
- Not statistically different
- Titration of PEEP to higher levels per protocol in the face mask group was limited due to patient intolerance and excess air leak
Strengths:
- RCT answering an important clinical question
- Randomization was blinded
- Consecutive enrollment
- Based on previous studies showing a 50% rate of intubation in patients with ARDS treated with NIV, the authors determined a 30% intubation rate (20% reduction) would be a clinically significant improvement
- Used an intention-to-treat analysis which extrapolates to real world practice
- Patients who died received a score of zero for ventilator-free days
- No patients were lost to follow up
- Patients well balanced between groups in terms of baseline characteristics
Limitations:
- Single center study with small number of patients may limit external validity
- Trial stopped early (Would a larger trial have shown as big a difference in the results?)
- Not possible to blind physicians and patients from helmet vs face mask NIV. This can have ramifications on a subjective outcome of need to intubate
- Patients not treated exactly the same:
- Median sustained PEEP (Helmet 8.0cmH20 vs Mask 5.1cmH20)
- FiO2 (Helmet 50% vs Mask 60%)
- Titration of PEEP to higher levels in face mask group was limited
- Secondary outcomes should be considered exploratory
- Trial not powered appropriately. Assuming an intubation rate of 50% of patients with hypoxemic respiratory failure requiring NIV, the study would have needed 206 patients to provide 90% power to detect a 20% absolute reduction of the primary outcome
- Like any new device, there is likely to be a learning curve as clinicians gain familiarity. This will require training of all physicians and staff (See Expert Opinion)
- This was a spectrum of illnesses causing ARDS, not an isolated diagnosis
Discussion:
- Helmet NIV:
- To avoid carbon dioxide rebreathing, pressure support set to maintain ventilator inspiratory flow rate > 100L/min
- To minimize inspiratory effort and optimize patient-ventilator synchrony, ventilator pressurization time set to 50ms and cycling off delay set to 50% of maximal inspiratory flow
- Decision to intubate based on predetermined criteria:
- Neurologic deterioration
- Persistent or worsening respiratory failure (i.e. O2 sat <88%, RR > 36/min)
- Intolerance of face mask or helmet
- Airway bleeding
- Copious respiratory secretions
- Authors chose an 8-hour period of face mask NIV a priori as a study entry criterion to avoid patients needing NIV for only a short time
- Given the risk of aerosolization is lower with helmet NIV it is important to point out that in one of the secondary exploratory analyses’ patients randomized to helmet group had substantially higher levels of PEEP, which was sustained throughout NIV. This also extrapolated to reductions in respiratory rate and similar oxygen saturation levels on a lower FiO2 than achieved with the face mask
- One important caveat to helmet NIV is that it has a large internal volume and high compliance which may lead to CO2 rebreathing. I wonder if this is the reason we see more neurologic issues such as AMS compared to face mask NIV
As I have never used a helmet NIV device I reached out to a friend in Italy, who has quite a bit of experience with these devices, for his thoughts…
Expert Opinion

Roberto Cosentini
Head Centro EAS Director – ASST Papa Giovanni XXIII – Bergamo, Italy
ASST Papa Giovanni XXIII – Bergamo, Italy
Milan, Italy
Twitter: @rob_cosentini
Staff:
- NIV (PSV) with the helmet is very difficult, because you have to pressurize a huge interface (15-20L hood) and while maintaining optimal synchronization. This means trouble fine-tuning the ventilator and NIV-specific monitoring (synchrony and hood tension).
- Furthermore, the hood is so big that tidal volume monitoring is not possible, which is important information of the disease course.
Equipment:
- The hood large volume needs a high performance (ICU) ventilator, since pressurization must be achieved in milliseconds.
- In my view, these limit the generalizability of the results
Author Conclusion: “Among patients with ARDS, treatment with helmet NIV resulted in a significant reduction of intubation rates. There was also a statistically significant reduction in 90-day mortality with helmet NIV. Multicenter studies are needed to replicate these findings.”
Clinical Take Home Point: Although this is an underpowered, single center study, there appears to be a signal that helmet NIV significantly reduces intubation rates among patients with ARDS compared to face mask NIV. Additionally, helmet NIV is associated with improved ventilator-free days, reduced ICU length of stay, as well as reduced 90d mortality, but these findings are only exploratory at this time. In lieu of a larger study, it seems that helmet NIV is a viable option and one that should be explored in patients with ARDS.
References:
- Patel BK et al. Effect of Noninvasive Ventilation Delivered by Helmet vs Face Mask on the Rate of Endotracheal Intubation in Patients with Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA 2016. PMID: 27179847

